Citizens Commission on Human Rights International [1]
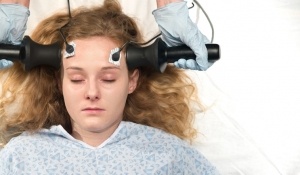
The Food and Drug Administration (FDA) has allowed the electroshock therapy (ECT) device to remain on the market without requiring clinical studies proving safety and efficacy, and is considering the American Psychiatric Association’s written proposal to expand ECT’s use on the general public, including on children.
According to the FDA’s mission statement, the FDA “is responsible for protecting the public health by ensuring the safety” and “efficacy” of medical products, including “medical devices” (the ECT device included).
Due to the documented risks attributed to ECT, we are calling on the FDA to prohibit its use.

10 Facts You May Not Know About Electroshock (ECT)
by Citizen’s Commission on Human Rights International [2]
The Mental Health Watchdog [2]
Psychiatry: Still Shocking After All These Years
1. FIRST CAME THE PIGS

The “science” behind electroshock as treatment was born out of a slaughterhouse in Rome, Italy. In 1938, Italian psychiatrist Ugo Cerletti saw pigs being electric shocked before slaughter, causing them to convulse and become more docile, thereby making them easier to kill. This inspired Cerletti to try the same “treatment” on humans. [1]
2. A “GOOD KIND OF BRAIN DAMAGE”?

Seventy-nine years after electroshock (Electroconvulsive Therapy or ECT) was invented, psychiatrists admit they still don’t know how it “works.” Numerous theories have been proposed, none of which have found mainstream scientific acceptance. Among the many psychiatric theories is that ECT “caused a good kind of brain damage.”[2]
3. NO LESS BRUTAL THAN IT USED TO BE

Psychiatrists claim that modern ECT is nowhere near as barbaric as the electroshock treatment depicted in the Academy-Award-winning film, One Flew Over the Cuckoo’s Nest. But the voltage and electrical current of modern ECT is higher than it was during the era of electroshock portrayed in the film, when patients used to break bones due to the violent convulsions produced by the “treatment.”[3]
4. WHAT’S “NEW” IS NOT IMPROVED

The reason patients no longer break bones isn’t because there is less electrical current (modern ECT uses a stronger current), but because muscle relaxants are first administered, making ECT appear less brutal. The muscle relaxant itself can be highly dangerous, with the most commonly used ECT muscle relaxant documented to cause cardiac arrest, prolonged respiratory muscle paralysis and potentially life-threatening and/or fatal allergic reactions.[4]
5. THE POWER TO HARM

Modern electroshock treatment delivers a pulse of current to the brain that is 7.5 times stronger than electrical fences used to deter bears, which deliver an electric shock to the bear when it touches the charged wires.[5] Yet psychiatrists are currently lobbying to increase the use of ECT on children.
6. THE APA WANTS MORE POWER TO SHOCK KIDS

The American Psychiatric Association (APA) has requested that the Food and Drug Administration (FDA), which regulates the ECT device, allow them to broaden their use of electroshock on children and adolescents. In a letter to the FDA, the APA states, “Having access to a rapid and effective treatment such as ECT is especially meaningful in children and adolescents….”[6]
7. ALREADY SHOCKING 0-5 YEAR-OLDS

Psychiatrists claim children are “infrequently” electroshocked, but no state or federal agency is documenting how many children are being electroshocked each year. State Medicaid records obtained solely through Freedom of Information Act (FOIA) requests have revealed psychiatrists are currently electroshocking children between the ages of 0 and 5 in the United States under the guise of “treatment.”[7]
8. COMMON SENSE – DON’T SHOCK KIDS

The World Health Organization’s Resource Book on Mental Health, Human Rights and Legislation states: “There are no indications for the use of ECT on minors, and hence this should be prohibited through legislation.” Four U.S. states and Western Australia have already banned ECT for those up to the ages of 12–16.[8]
9. USED ON PREGNANT WOMEN

Electroshock is also being used on pregnant women, even in the third trimester. While psychiatrists claim this is perfectly safe, a study done by Norwegian researchers found the infant mortality rate was 7.1% following the administration of ECT to pregnant mothers. Documented adverse events of ECT on pregnant women include miscarriage, stillbirth, fetal heart problems and malformations. [9]
10. NO SAFETY OR EFFICACY REQUIRED

The only mode of “regulation” for the use of electroshock in the United States is the FDA, which regulates the device. The FDA has never required any clinical trials proving safety or efficacy of the treatment. It has simply allowed the device to remain on the market, despite admitting electroshock can cause permanent memory loss, cardiovascular complications, brain damage and death. [10]
Summary: Most people don’t believe that electroshock (electroconvulsive therapy or ECT) is still being used in modern society. This is most likely because the mere idea of electroshock is so abhorrent that most people simply can’t believe it’s still being used. Factually, ECT is one of the most brutal “treatments” ever inflicted upon individuals under the guise of mental health care. Yet approximately one million people worldwide, including an estimated 100,000 Americans, are given electroshock each year, including the elderly, pregnant women and children.
To support the campaign to Ban Electroshock, click here [1].
Read the full article at CCHR.org. [2]
REFERENCES:
[1] “Origins of ECT,” Convulsive Therapy, Vol. 4., No. 1., 1988, p. 7,
[2] Aaron Levin, “ECT Said to Be Valid Alternative For Elderly Patients With Depression,” Psychiatric News, 3 May 2016,
[3] Douglas G. Cameron, “ECT: Sham Statistics, the Myth of Convulsive Therapy, and the Case for Consumer Misinformation,” The Journal of Mind and Behavior, Vol 15, No 1 and 2, Winter and Spring 1994, pp 177-198,
[4] Anectine ® (Succinylcholine Chloride Injection, USP) Label, FDA.gov.,
[5] Somatics Thymatron® System IV Brochure, Somatics LLC, /somatics_brochure.pdf [3], states that the ECT device uses a current of 0.9 amps (or 900 milliamps); “Are your bear systems dangerous?,” Tahoe Bear Busters, http
s://www.tahoebearbusters.com/?faqs=arebearsystemsdangerous, states that electric fences average around 120 milliamps.
[6] Letter to Robert M. Califf, M.D., Commissioner, U.S. Food and Drug Administration, from the American Psychiatric Association, March 10, 2016, https://psychiatry.org/File%20Library/Psychiatrists/Advocacy/Federal/APA-FDA-ECT-reclassification-comments-03102016.pdf. [4]
[7] State records obtained through public records requests from Utah, Indiana, Oregon and Kentucky.
[8] “WHO RESOURCE BOOK ON MENTAL HEALTH, HUMAN RIGHTS AND LEGISLATION,” World Health Organization, 2005, p. 64.
[9] Kari Ann Leiknes, et al. “Electroconvulsive therapy during pregnancy: a systematic review of case studies,” Arch Womens Ment Health, epub 24 Nov 2013,
[10] NEUROLOGICAL DEVICES PANEL, CENTER FOR DEVICES AND RADIOLOGICAL HEALTH MEDICAL DEVICES ADVISORY COMMITTEE, FOOD AND DRUG ADMINISTRATION, pp 148-149, 27 Jan 2011, http://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/MedicalDevicesAdvisoryCommittee/neurologicalDevicesPanel/UCM247594.pdf.%20Accessed%20December%203; “Electroconvulsive Therapy (ECT) Devices for Class II Intended Uses,” Draft Guidance for Industry, Clinicians and Food and Drugs Administration Staff, Food and Drug Administration, Center for Devices and Radiological Health, 29 Dec 2015, https://www.fda.gov/downloads/MedicalDevices/…/UCM478942.pdf. [5]